 /Radium226-56a12a7a5f9b58b7d0bcac85.jpg)
Using a primitive X-ray machine, the physician discovered serious bone decay, the likes of which he had never seen. Her troubles were compounded when her jaw became swollen and inflamed, so she sought the assistance of a doctor in diagnosing the inexplicable symptoms. All rights reserved.In 1922, a bank teller named Grace Fryer became concerned when her teeth began to loosen and fall out for no discernible reason. Copyright © 2022, Columbia University Press. The Columbia Electronic Encyclopedia, 6th ed. Metallic radium was isolated by electrolysis in 1910 by Marie Curie and André Debierne they first formed a mercury-radium amalgam by electrolysis and then removed the mercury by distillation. They had earlier found polonium in a similar sample. Radium was discovered in 1898 by Pierre and Marie Curie in pitchblende given them by Austria after the uranium salts had been removed for use in glass manufacture. Other radioisotopes (e.g., cobalt-60) are often used in its place when they are less expensive, more powerful, or safer to use. Radium also is a dangerous material prolonged exposure to even small amounts may cause cancer, anemia, or other disorders. The small amount of the element present in any ore and the difficulty of extraction make it expensive. It is recovered as the bromide by an involved chemical process. 
Radium is usually obtained (with barium impurities) in residues from the production of uranium. Radium is present in all uranium minerals and is widely distributed in small amounts. Although some radium is obtained from carnotite from Colorado, the chief sources are carnotite from Congo (Kinshasa) and pitchblende from W Canada. 
Its compounds are found in uranium ores there is usually about 1 part of radium to 3 million parts of uranium in these ores. Radium decreases in radioactivity about 1% in 25 years. The curie is a unit of radioactivity defined as that amount of any radioactive substance that has the same disintegration rate as 1 gram of radium-226, i.e., 3.7×10 10 disintegrations per sec. In its radioactive decay radium emits alpha, beta, and gamma rays and also produces heat (about 1,000 calories per gram per year). It is a product in the radioactive decay series of uranium-238 it is immediately preceded in this series by thorium-230 and followed by radon-222 (a gas formerly called radium emanation). Sixteen isotopes of radium are known, but only radium-226 (half-life 1,599 years), the most stable of the isotopes, is used commercially. Radium is also used as a neutron source (mixed with beryllium) and as a gamma-ray source. Mixed with a phosphor such as zinc sulfide, radium compounds are used in luminous paints. 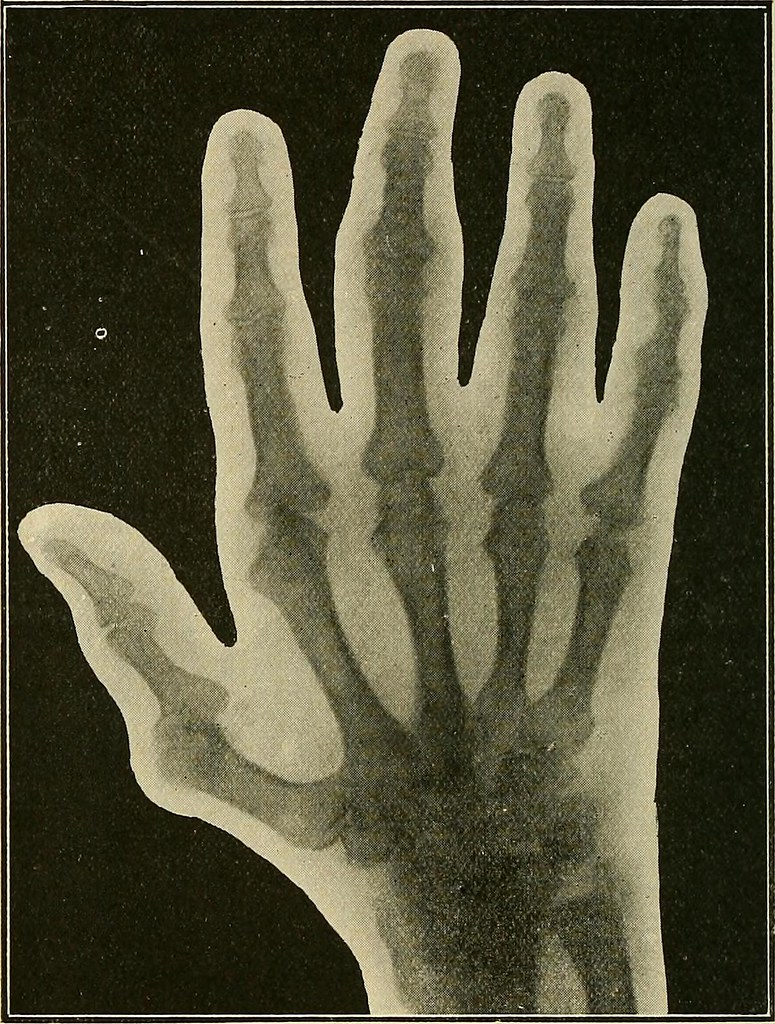
The most important property of radium and its compounds is their radioactivity radiotherapy is used in medicine in the treatment of cancer. It reacts with acids to form the commercially important chloride and bromide. It combines directly with water to form the hydroxide. When radium is exposed to air, a black coating of nitride rapidly forms. It is an alkaline-earth metal in its chemical properties it closely resembles barium, the element above it in Group 2 of the periodic table. Radium is a lustrous white radioactive metal. 88 mass number of most stable isotope 226 m.p. Radium rā´dēəm, radioactive metallic chemical element symbol Ra at. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
